Making an explosive gas out of sand, magnesium and drain cleaner
As you’ve probably noticed from my previous articles, I really enjoy making things light up, whether it’s through fluorescence, chemiluminescence, or simply causing them to explode. Today, I’ll share with you how my friend Máximo and I made a gas that does the job for you: it explodes on its own, it’s pyrophoric! Combining a few easily accessible materials, we synthesised silane (SiH4), the analogue of methane (CH4) that ignites and explodes upon contact with air.

Chemistry is cooking
In a beaker, we added a handful of magnesium powder and a few pinches of sand, using a rather unscientific approach. We mixed them up and put them in a boiling tube, covering the mixture with a bit of extra sand to prevent pesky oxygen (O2) from oxidising our magnesium. We then put it on a clamp stand outside (somewhere no one could see us) and placed a Bunsen burner beneath it. We lit up the gas and walked a few metres back.
It got scary. Smoke started seeping out, the sand-magnesium mixture turned black, and we heard glass cracking. In case it exploded, we took a long branch and launched the Bunsen burner far away, then ran to turn off the flame before our school went down in flames. I guess we were simultaneously crazy scientists and pyromaniac baseball players scoring a home run.
After waiting for the threat to subside, we approached the boiling tube, wrapped it in paper, and broke it with a stone, releasing the shiny crystals formed inside.

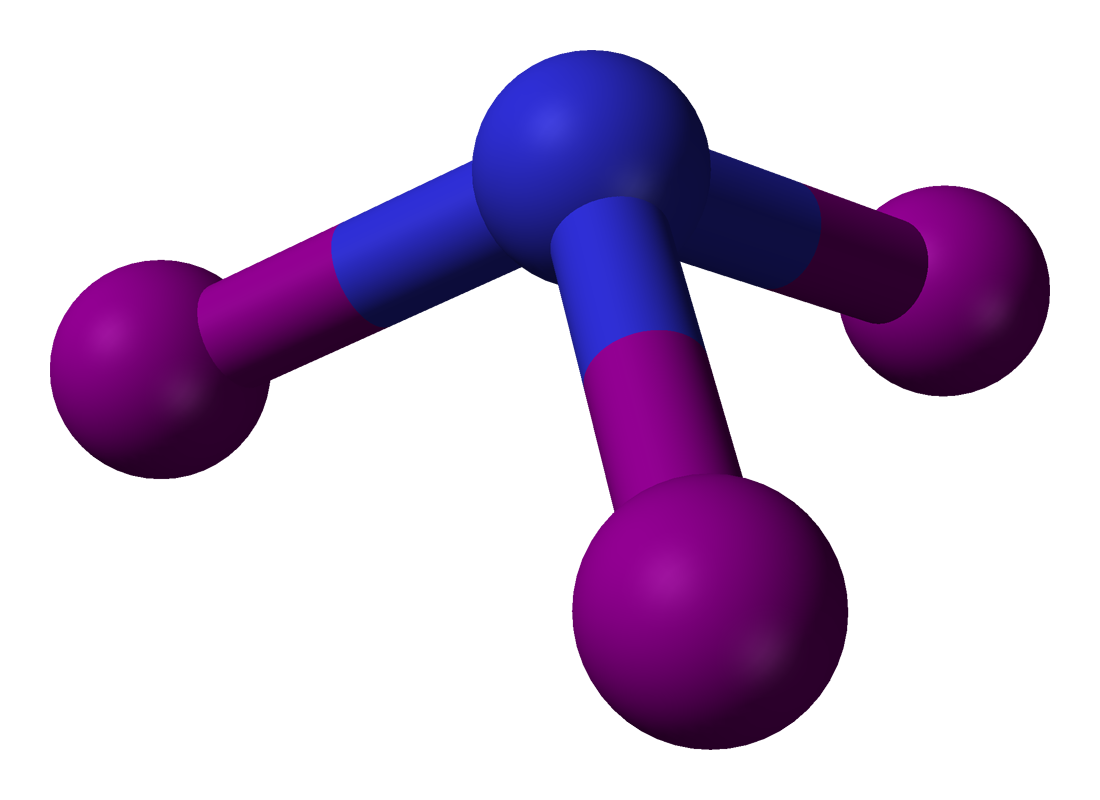
In this process, amorphous silicon (Si) and then magnesium silicide (Mg2Si) are formed via the following reactions:
1. SiO2(s) + 2Mg(s) → Si(s) + 2MgO(s)
2. 2Mg(s) + Si(s) → Mg2Si(s)
Magnesium silicide is the compound that forms the black-violet crystals pictured above.

We took the magnesium silicide crystals and ground them up in a mortar and pestle to increase their surface area. Then, we prepared a beaker containing a pint of dilute hydrochloric acid (HCl) and placed it on the magnetic stirrer in our darkened fume hood. To make silane, we added the magnesium silicide powder to the hydrochloric acid, producing the following reaction:
Mg2Si(s) + 4HCl(aq) → 2MgCl2(aq) + SiH4(g)
The silane bubbles out of solution, and when it comes in contact with the air… BOOM! This is one of the reactions where silane explodes upon coming in contact with air:
SiH4(g) + 2O2(g) → SiO2(s) + 2H2O(g)
A whirlpool of fireworks in a beaker of drain cleaner
To get the best views of this spectacle, you can watch my video in full screen.